Bromomethane
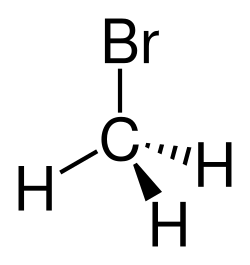
Bromomethane, also known as methyl bromide, is an organobromine compound with the chemical formula CH₃Br. It is a colorless, nonflammable gas with a slightly sweet odor, and it is used primarily as a fumigant for soil and agricultural products.
Chemical Properties[edit]
Bromomethane is a halomethane and is classified as a methyl halide. It is a simple alkyl halide, consisting of a methyl group (CH₃) bonded to a bromine atom. The compound is volatile and has a boiling point of 3.6 °C. It is slightly soluble in water but more soluble in organic solvents.

Production[edit]
Bromomethane is produced both naturally and synthetically. In nature, it is produced by marine organisms such as kelp, algae, and phytoplankton. Industrially, it is synthesized by the reaction of methanol with hydrogen bromide or by the reaction of methane with bromine in the presence of a catalyst.
Uses[edit]
Bromomethane is primarily used as a fumigant to control pests in agriculture. It is effective against a wide range of pests, including insects, nematodes, and weeds. It is also used in the quarantine and pre-shipment treatment of agricultural commodities.

Environmental Impact[edit]
Bromomethane is a significant ozone-depleting substance. Its use is regulated under the Montreal Protocol, which aims to phase out substances that deplete the ozone layer. As a result, its use has been significantly reduced, and alternatives are being sought.
Health Effects[edit]
Exposure to bromomethane can be harmful to human health. It can cause respiratory distress, neurological effects, and skin irritation. Safety measures are essential when handling this chemical to prevent exposure.

Spectroscopy[edit]
Bromomethane can be analyzed using infrared spectroscopy, which provides information about its molecular vibrations and structure. The IR spectrum of bromomethane shows characteristic absorption bands that can be used to identify the compound.

Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian