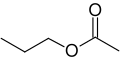
Propyl group
Propyl group is a functional group in organic chemistry. It is an alkyl group with the chemical formula -C3H7. The propyl group is derived from propane by removing one hydrogen atom from the end of the chain.
Structure and Properties[edit]
The propyl group can exist in two different forms, or isomers. The straight-chain form is known as n-propyl or normal propyl. The branched form, where the carbon atoms are not all in a straight line, is known as iso-propyl.
The propyl group is a nonpolar molecule, meaning it does not have a net electric charge. This makes it hydrophobic, or water-repelling. It is also relatively small, which allows it to fit into many different chemical structures.
Uses and Applications[edit]
Propyl groups are commonly used in organic synthesis. They can be added to other molecules to increase their size, change their reactivity, or introduce new functional groups.
In the pharmaceutical industry, propyl groups are often used to modify the properties of drugs. For example, the addition of a propyl group to a molecule can increase its lipophilicity, helping it to cross cell membranes more easily.
In the food industry, propyl groups are found in many artificial flavors and fragrances. They can also be used to make certain types of plastic and rubber.
See Also[edit]
References[edit]
-
Propyl groups
-
Propyl acetate
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian