Epothilone
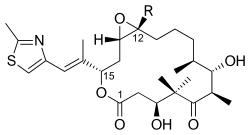
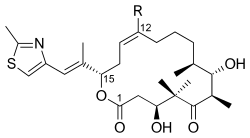
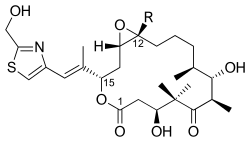
Epothilone is a class of macrocyclic lactones that are produced by the bacteria Sorangium cellulosum. These compounds are of significant interest in the field of biotechnology and pharmacology due to their potent antimitotic activity, which is similar to that of taxanes, a well-known class of chemotherapy agents used in the treatment of cancer. Epothilones work by stabilizing microtubules, thereby inhibiting cell division, which makes them effective in the treatment of various malignancies.
History[edit]
Epothilones were first isolated in the early 1990s from the myxobacterium Sorangium cellulosum. The discovery of epothilones arose from the search for new compounds with antimitotic activity that could serve as alternatives to taxanes, which are derived from the bark of the Pacific yew tree and have limited availability. The unique mechanism of action and the ability to synthesize epothilones in the laboratory made them a promising candidate for development into anticancer drugs.
Mechanism of Action[edit]
Epothilones exert their anticancer effects by binding to beta-tubulin subunits of microtubules. This binding leads to the stabilization of microtubules, preventing their depolymerization. As a result, the mitotic spindle cannot function properly during cell division, leading to cell cycle arrest at the G2/M phase and eventually cell death. This mechanism is similar to that of taxanes but with the advantage that epothilones can remain effective against taxane-resistant cancers due to differences in their binding sites or their ability to be transported into cells.
Clinical Use and Research[edit]
Several derivatives of epothilone, including ixabepilone and patupilone, have been developed and tested in clinical trials for their efficacy against various types of cancer, including breast cancer, ovarian cancer, and non-small cell lung cancer. Ixabepilone, in particular, has been approved by the Food and Drug Administration (FDA) for the treatment of aggressive and metastatic breast cancer, especially in patients who are resistant to anthracyclines and taxanes.
Chemical Structure and Synthesis[edit]
Epothilones are characterized by their macrocyclic lactone structure, which is essential for their biological activity. The complexity of their structure has made the synthesis of epothilones a challenging task for chemists. However, advances in synthetic techniques have allowed for the production of epothilone derivatives with improved pharmacological properties, such as increased solubility and stability, making them more suitable for clinical use.
Future Directions[edit]
Research on epothilones continues to explore their potential as anticancer agents, with studies focusing on understanding their mechanism of action, overcoming resistance mechanisms, and improving their pharmacokinetic profiles. The development of novel epothilone derivatives with enhanced efficacy and reduced toxicity is an ongoing area of interest, which holds promise for the future of cancer therapy.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian