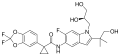
Elexacaftor/tezacaftor/ivacaftor
Elexacaftor/tezacaftor/ivacaftor is a combination drug used in the treatment of cystic fibrosis (CF) in individuals aged 12 years and older who have at least one F508del mutation in the CFTR gene, which is the most common mutation causing cystic fibrosis. This combination therapy represents a significant advancement in the management of cystic fibrosis, targeting the underlying cause of the disease rather than just its symptoms.
Composition[edit]
The drug is a combination of three different molecules: elexacaftor, tezacaftor, and ivacaftor. Each component plays a unique role in enhancing the function of the defective CFTR protein found in individuals with cystic fibrosis.
- Elexacaftor works by increasing the amount of CFTR protein on the cell surface.
- Tezacaftor facilitates the movement of the CFTR protein to the cell surface.
- Ivacaftor increases the activity of the CFTR protein once it is on the cell surface.
Mechanism of Action[edit]
The CFTR protein is essential for regulating salt and water transport across cell membranes, which is crucial for the production of mucus, sweat, saliva, tears, and digestive enzymes. Mutations in the CFTR gene lead to the production of a defective CFTR protein, resulting in the thick, sticky mucus characteristic of cystic fibrosis. The combination of elexacaftor, tezacaftor, and ivacaftor addresses the defective CFTR protein's function by improving its quantity and activity on the cell surface, thereby alleviating the symptoms of cystic fibrosis.
Clinical Use[edit]
This combination therapy is indicated for the treatment of cystic fibrosis in patients with specific genetic mutations. It has shown significant improvements in lung function, nutritional status, and overall quality of life in individuals with cystic fibrosis. However, it is important to note that this therapy is not a cure for cystic fibrosis but a treatment that manages the condition.
Side Effects[edit]
Common side effects include headache, upper respiratory tract infection, abdominal pain, diarrhea, rash, and flu-like symptoms. Liver function tests should be monitored regularly during treatment, as the therapy can affect liver enzymes.
Approval[edit]
The combination of elexacaftor/tezacaftor/ivacaftor was approved by the Food and Drug Administration (FDA) in the United States and by the European Medicines Agency (EMA) in the European Union. Its approval marked a significant milestone in the treatment of cystic fibrosis, offering hope to many patients and families affected by this challenging condition.
See Also[edit]
Elexacaftor/tezacaftor/ivacaftor[edit]
-
Mechanism of action of Elexacaftor/tezacaftor/ivacaftor
-
Complex of Ivacaftor bound to CFTR
-
Ivacaftor structure
-
Elexacaftor structure
-
Tezacaftor structure
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian