Baeyer–Villiger oxidation
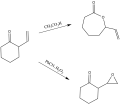
Baeyer–Villiger Oxidation is an organic reaction that involves the oxidative insertion of an oxygen atom into a carbon-carbon bond adjacent to a carbonyl, which results in the conversion of ketones to esters and cyclic ketones to lactones. This reaction is named after the German chemists Adolf von Baeyer and Victor Villiger who discovered it in 1899. The Baeyer–Villiger oxidation is a valuable transformation in organic synthesis due to its ability to introduce oxygen functionalities into molecules, which can be further manipulated for the synthesis of complex organic compounds.
Mechanism[edit]
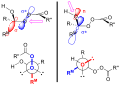
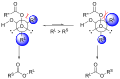
The mechanism of the Baeyer–Villiger oxidation involves several key steps. Initially, the ketone undergoes nucleophilic attack by a peroxide, such as mCPBA (meta-chloroperoxybenzoic acid) or hydrogen peroxide, to form a 'Criegee intermediate'. This intermediate is a tetrahedral structure where the oxygen of the peroxide is inserted between the carbonyl carbon and the adjacent carbon atom. Subsequently, a rearrangement occurs, which is facilitated by the migration of an alkyl or aryl group from the carbon adjacent to the oxygen to the carbonyl carbon, leading to the formation of an ester or lactone. The migratory aptitude of groups in this rearrangement typically follows the order: tertiary alkyl > secondary alkyl > primary alkyl > methyl.
Applications[edit]
The Baeyer–Villiger oxidation has found widespread application in organic synthesis, including the synthesis of pharmaceuticals, agrochemicals, and fragrances. It is particularly useful in the synthesis of lactones, which are core structures in many natural products and biologically active molecules. Additionally, the reaction has been employed in the preparation of polymers and materials science.
Variants[edit]
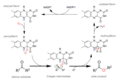
Several variants of the Baeyer–Villiger oxidation exist, which employ different oxidizing agents and conditions. For example, the use of hydrogen peroxide as an oxidant in the presence of a catalytic amount of a selenium or vanadium compound allows for a more environmentally friendly process. Furthermore, biocatalytic approaches using enzymes such as Baeyer–Villiger monooxygenases have been developed, offering highly selective and mild conditions for the oxidation.
Limitations[edit]
Despite its utility, the Baeyer–Villiger oxidation has some limitations. The reaction generally requires stoichiometric amounts of the oxidizing agent, which can lead to waste disposal issues. Additionally, the reaction can be sensitive to the substrate structure, and the migratory aptitude of different groups can limit the scope of the reaction.
Conclusion[edit]
The Baeyer–Villiger oxidation is a fundamental reaction in organic chemistry that enables the efficient synthesis of esters and lactones from ketones. Its versatility and broad applicability have made it an essential tool in the synthesis of complex organic molecules.
Baeyer–Villiger oxidation gallery[edit]
-
Baeyer-Villiger oxidation
-
Baeyer-Villiger oxidation reaction mechanism
-
Baeyer-Villiger oxidation stereoelectronic effects
-
Criegee intermediate resonance structures
-
Criegee intermediate stereoelectronics
-
Original Baeyer-Villiger oxidation reactions
-
Proposed Baeyer-Villiger Intermediates
-
Dorfman and Doering's Labelling Experiment
-
Reagent Dependent Oxidation
-
BVMO reaction mechanism
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
- Budget GLP-1 shots
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian