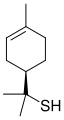
Grapefruit mercaptan
Grapefruit Mercaptan[edit]
Grapefruit mercaptan
```
This template is designed for creating an infobox for chemical compounds on a MediaWiki-based site, similar to those found on WikiMD. It includes fields for the compound's name, image, IUPAC name, other names, CAS number, PubChem ID, ChemSpider ID, UNII, KEGG ID, ChEBI ID, molecular formula, molar mass, appearance, density, melting point, boiling point, solubility in water, SMILES and InChI chemical structure identifiers, EU Index, EC Number, RTECS number, LD50, and related compounds. The template also includes a stub notice for chemistry-related articles, encouraging users to expand the article.
Grapefruit Mercaptan is a chemical compound with the IUPAC name 1,8-dimercapto-p-menthane. It is commonly used in the fragrance industry due to its unique odor profile. Grapefruit Mercaptan is known for its strong, fruity, and slightly sulfurous aroma, reminiscent of grapefruit.
Properties[edit]
Grapefruit Mercaptan is a colorless liquid with a density of 0.97 g/cm3. It has a molar mass of 204.39 g/mol. The compound has a melting point of -30 °C and a boiling point of 180 °C. Grapefruit Mercaptan is insoluble in water.
Uses[edit]
Grapefruit Mercaptan is primarily used as a fragrance ingredient in perfumes, colognes, and other scented products. Its distinct grapefruit-like scent adds a refreshing and uplifting note to various personal care and household products.
Safety Precautions[edit]
Grapefruit Mercaptan is flammable and should be handled with care. It has a flash point of 60 °C and an autoignition point of 230 °C. Proper safety measures, such as using appropriate protective equipment and storing the compound in a well-ventilated area, should be followed when working with Grapefruit Mercaptan.
References[edit]
-
Grapefruit mercaptan
-
Grapefruit mercaptan 3D balls
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian