Alpha helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
-
Alpha_helix
Alpha Helix[edit]
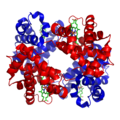
The alpha helix is a common structural motif in the secondary structure of proteins. It is characterized by a right-handed coil where each amino acid residue corresponds to a 100-degree turn in the helix, and 3.6 residues per turn. The alpha helix is stabilized by hydrogen bonds between the backbone amide hydrogen of one amino acid and the carbonyl oxygen of another amino acid four residues earlier.
Structure[edit]
The alpha helix is a right-handed spiral conformation in which every backbone N-H group forms a hydrogen bond with the backbone C=O group of the amino acid located four residues earlier along the protein sequence. This pattern of hydrogen bonding confers significant stability to the helical structure. The side chains of the amino acids in an alpha helix extend outward from the helical backbone, allowing them to interact with other molecules or parts of the protein.
The pitch of the alpha helix, which is the vertical distance between turns, is approximately 5.4 Ångströms. The helical structure is compact, with a diameter of about 10 Ångströms, and it is often found in the hydrophobic core of globular proteins.
Formation[edit]
The formation of an alpha helix is influenced by the amino acid sequence of the polypeptide. Certain amino acids, such as alanine, leucine, and methionine, are more likely to be found in alpha helices due to their favorable interactions and steric properties. In contrast, amino acids like proline and glycine are less common in alpha helices; proline introduces a kink in the helix due to its rigid ring structure, while glycine is too flexible and disrupts the helical structure.
Function[edit]
Alpha helices play a crucial role in the function of many proteins. They can form coiled-coil structures, which are important for the structural integrity of fibrous proteins such as keratin and myosin. In membrane proteins, alpha helices often span the lipid bilayer, forming channels or pores that allow the passage of ions and small molecules. The amphipathic nature of alpha helices, with hydrophilic and hydrophobic sides, allows them to interact with both the aqueous environment and the hydrophobic core of the membrane.
Discovery[edit]
The concept of the alpha helix was first proposed by Linus Pauling, Robert Corey, and Herman Branson in the early 1950s. Their work on the structure of proteins laid the foundation for understanding protein folding and function. The alpha helix was one of the first secondary structures to be identified, and it remains a fundamental concept in the study of protein structure.
Related Pages[edit]


Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian