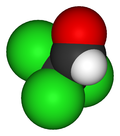
Chloral
Chloral, also known as trichloroacetaldehyde or CCl3CHO, is an organic compound with significant historical and industrial relevance. It is a colorless liquid with a pungent odor and was first synthesized in the early 19th century. Chloral is primarily known for its role in the synthesis of chloral hydrate, a sedative and hypnotic drug.
History[edit]
Chloral was first synthesized by the chemist Justus von Liebig in 1832 through the chlorination of ethanol. This discovery marked a significant milestone in organic chemistry, illustrating the transformation of a simple organic molecule into a more complex one through halogenation.
Chemical Properties[edit]
Chloral is a haloaldehyde, characterized by the presence of three chlorine atoms and an aldehyde group attached to the same carbon atom. It is typically a dense, colorless liquid that is soluble in a variety of organic solvents. Chloral tends to hydrolyze in the presence of water to form chloral hydrate, which is more stable and has been widely used in medicine.
Synthesis[edit]
The primary method for synthesizing chloral involves the chlorination of ethanol, which can be conducted using either chlorine gas or a mixture of calcium hypochlorite (bleaching powder) and acetic acid. This reaction produces chloral along with byproducts such as hydrochloric acid and calcium chloride.
Uses[edit]
Chloral's most notable application is in the production of chloral hydrate, which was historically used as a sedative and hypnotic before the development of safer alternatives. Chloral can also undergo further chemical reactions to produce DDT (dichlorodiphenyltrichloroethane), an insecticide that was widely used until concerns about its environmental and health impacts led to widespread bans.
Safety and Environmental Impact[edit]
Chloral is considered a hazardous material due to its potential health effects, which include irritation to the skin, eyes, and respiratory system. It is also a precursor to DDT, which has significant environmental impacts, including bioaccumulation and effects on wildlife. Proper handling and disposal of chloral are essential to minimize these risks.
Conclusion[edit]
While chloral itself is not as widely used today, its derivatives, particularly chloral hydrate, have played a significant role in medicine and chemistry. The compound's reactivity and transformation into various industrially and pharmacologically important compounds continue to make it a subject of study in organic chemistry.
Chloral[edit]
-
Chloral 2D Skeletal
-
Chloral 3D van der Waals
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian