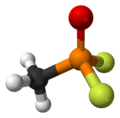
Methylphosphonyl difluoride
Methylphosphonyl difluoride (MPDF) is a chemical compound with the formula CH3POF2. It is a colorless, volatile liquid that is primarily used in the synthesis of various chemical agents, including some of the most potent nerve agents known, such as Sarin (GB) and Soman (GD). Due to its use in the production of chemical weapons, MPDF is listed under Schedule 1 of the Chemical Weapons Convention (CWC), making its production and stockpiling highly regulated and monitored internationally.
Properties
Methylphosphonyl difluoride is characterized by its high reactivity, especially with compounds containing active hydrogen atoms, such as water and alcohols. This reactivity is due to the presence of the phosphonyl (PO) and difluoride (F2) groups, which make it an effective intermediate in the synthesis of organophosphorus compounds. MPDF is relatively stable at room temperature but decomposes upon contact with moisture, releasing hydrofluoric acid (HF), a highly corrosive and toxic substance.
Synthesis
The synthesis of methylphosphonyl difluoride typically involves the reaction of dimethyl methylphosphonate with hydrogen fluoride (HF) under specific conditions. This process requires careful handling due to the highly corrosive nature of HF and the toxic properties of MPDF itself.
Applications
While the primary use of MPDF has been in the production of nerve agents, research into less harmful applications is ongoing. Due to its ability to form stable organophosphorus compounds, MPDF has potential uses in various industrial and pharmaceutical applications. However, its association with chemical weapons significantly limits its availability and use in research and industry.
Health Effects
Exposure to methylphosphonyl difluoride can have severe health effects, including respiratory distress, skin and eye irritation, and, if ingested or inhaled in sufficient quantities, it can be fatal. The compound's ability to release hydrofluoric acid upon decomposition adds to its hazardous nature, as HF is highly toxic and can cause deep tissue damage and systemic toxicity.
Regulation and Monitoring
Due to its potential use in the production of chemical weapons, the production, storage, and use of MPDF are strictly regulated under the Chemical Weapons Convention. Countries that are signatories to the CWC are required to declare and destroy any stockpiles of MPDF and are subject to inspections to ensure compliance with the treaty.
See Also
Methylphosphonyl_difluoride
-
Methylphosphonyl difluoride 2D structure
-
Methylphosphonyl difluoride 3D ball model
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian