Organoboron chemistry
Organoboron chemistry is the study of chemical compounds containing boron bonded to carbon, or organoboron compounds. This area of chemistry is important both in academic research and industrial applications because of the unique reactions and properties of organoboron compounds. Boron's trivalent nature allows it to form stable covalent bonds with carbon, making organoboron compounds versatile intermediates in organic synthesis, particularly in the formation of carbon-carbon bonds.
Overview[edit]
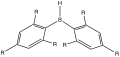
Organoboron chemistry involves the synthesis, characterization, and application of organoboron compounds. Boron, a metalloid element in group 13 of the periodic table, has a unique chemistry due to its small size, high electronegativity, and the ability to form stable compounds with carbon. The most common organoboron compounds are trialkylboranes and triarylboranes, but the field encompasses a wide range of compounds, including boronic acids, boronate esters, and borinic esters.
Synthesis[edit]
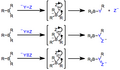
The synthesis of organoboron compounds can be achieved through several methods. One of the most common methods is the hydroboration reaction, where alkenes or alkynes are treated with borane (BH3) or its derivatives to form trialkylboranes. Other methods include the direct borylation of aryl and alkyl halides, transmetalation reactions where a metal-boron bond is formed by exchanging a metal from another metal-organic compound, and the use of diborane (B2H6) for the direct borylation of hydrocarbons.
Reactions[edit]
Organoboron compounds participate in a variety of chemical reactions, making them valuable tools in organic synthesis. One of the most important reactions is the Suzuki-Miyaura coupling, where a boronic acid or its ester reacts with an aryl or vinyl halide in the presence of a palladium catalyst to form a new carbon-carbon bond. Other notable reactions include the Miyaura borylation, which involves the direct borylation of aryl halides, and the Petasis reaction, a multicomponent reaction involving a boronic acid, an amine, and an aldehyde or ketone to form amines.
Applications[edit]
Organoboron compounds have a wide range of applications in organic synthesis, medicinal chemistry, and material science. In organic synthesis, they are used as intermediates in the synthesis of complex molecules, including pharmaceuticals, agrochemicals, and natural products. In medicinal chemistry, boron-containing drugs have been developed for the treatment of various diseases, such as cancer and infectious diseases. In material science, organoboron compounds are used in the development of organic light-emitting diodes (OLEDs) and as precursors for the synthesis of advanced materials.
Safety[edit]
The handling and use of organoboron compounds require caution due to their potential toxicity and reactivity. Safety measures include the use of appropriate personal protective equipment (PPE), working in a well-ventilated area or fume hood, and proper storage and disposal of organoboron compounds.
See also[edit]
References[edit]
<references />
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
-
Organoboron chemistry
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian