Density of states
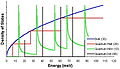
Density of States (DOS) refers to the number of states per interval of energy at each energy level that are available to be occupied by electrons, phonons, or other particles. It is a fundamental concept in solid-state physics, materials science, and quantum chemistry, providing crucial insights into the electronic structure and properties of materials. The DOS plays a vital role in determining the electrical, thermal, and optical properties of materials.
Overview[edit]
The concept of the density of states is particularly important in the study of semiconductors, metals, and insulators. It helps in understanding and predicting the behavior of electrons in a material under various conditions, such as in the presence of an electric field, magnetic field, or at different temperatures.
Mathematical Formulation[edit]
The density of states \(g(E)\) is defined as the number of states per unit volume per energy range. Mathematically, it can be expressed as: \[ g(E) = \frac{dN}{dE} \] where \(dN\) is the number of states in the energy range \(E\) to \(E + dE\).
For a three-dimensional system, the density of states for free electrons, considering the effective mass approximation, can be derived to be proportional to the square root of the energy: \[ g(E) \propto \sqrt{E} \]
Types of Density of States[edit]
There are several types of density of states, including:
- Local Density of States (LDOS): Refers to the DOS at a specific point in space, providing spatial resolution of the states.
- Partial Density of States (PDOS): Gives the contribution of each atomic orbital to the total DOS, useful in understanding the electronic structure of materials.
- Total Density of States (TDOS): The sum of the PDOS over all atoms and orbitals, representing the overall DOS of a system.
Significance in Material Science[edit]
The DOS is crucial in determining the electronic properties of materials. For instance, in semiconductors, the DOS at the valence band and conduction band edges determines the carrier concentration and hence the conductivity. In metals, the DOS at the Fermi level is directly related to the electronic specific heat and magnetic susceptibility.
Applications[edit]
- Photovoltaics: Understanding the DOS helps in designing materials with optimal band gaps for solar energy conversion.
- Thermoelectrics: Materials with a high DOS at specific energy levels can lead to enhanced thermoelectric performance.
- Superconductors: The DOS plays a key role in determining the critical temperature and other properties of superconductors.
Challenges and Future Directions[edit]
One of the challenges in utilizing the DOS concept is the accurate calculation and measurement of the DOS in complex materials. Advanced computational methods and spectroscopic techniques are continually being developed to address this challenge.
See Also[edit]
References[edit]
<references/>
-
Brillouin Zone (1st, FCC)
-
Dual Cube-Octahedron
-
K-space
-
Monoatomic chain phonon dispersion
-
Free-electron DOS
-
Fermi-Dirac product with Density of States
-
DOS multidimensional
-
Omen local density of states
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian