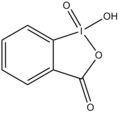
2-Iodoxybenzoic acid
2-Iodoxybenzoic acid[edit]

2-Iodoxybenzoic acid (IBX) is an organoiodine compound that is widely used as an oxidizing agent in organic chemistry. It is a hypervalent iodine compound, specifically a _5-iodane, and is known for its ability to selectively oxidize alcohols to aldehydes or ketones.
Structure and Properties[edit]
2-Iodoxybenzoic acid is characterized by the presence of an iodine atom in a +5 oxidation state, bonded to an aromatic ring. The iodine atom is also bonded to two oxygen atoms, forming an iodine-oxygen double bond and an iodine-oxygen single bond, which gives the compound its oxidizing properties.
Synthesis[edit]

2-Iodoxybenzoic acid is typically synthesized from 2-iodobenzoic acid through oxidation. The oxidation is often carried out using strong oxidizing agents such as sodium periodate or potassium bromate in an acidic medium.
Applications in Organic Synthesis[edit]
2-Iodoxybenzoic acid is primarily used as an oxidizing agent in organic synthesis. It is particularly effective in the oxidation of primary and secondary alcohols to aldehydes and ketones, respectively.
Oxidation of Alcohols[edit]

IBX is known for its mild and selective oxidation of alcohols. It can oxidize primary alcohols to aldehydes without over-oxidizing them to carboxylic acids. Secondary alcohols are oxidized to ketones.
Oxidative Cleavage[edit]

IBX can also be used for the oxidative cleavage of vicinal diols, leading to the formation of two carbonyl compounds. This reaction is useful in the degradation of complex molecules into simpler fragments.
Use in Dimethyl Sulfoxide (DMSO)[edit]

In the presence of dimethyl sulfoxide (DMSO), IBX can facilitate the oxidative cleavage of certain substrates, providing a versatile method for the transformation of organic compounds.
Safety and Handling[edit]
2-Iodoxybenzoic acid is a powerful oxidizing agent and should be handled with care. It is important to use appropriate personal protective equipment and to work in a well-ventilated area to avoid exposure to its potentially harmful effects.
Related pages[edit]
Gallery[edit]
-
Example of IBX oxidation
2-Iodoxybenzoic_acid[edit]
-
2-Iodoxybenzoic acid structure
-
Preparation of 2-Iodoxybenzoic acid
-
IBX oxidation mechanism
-
Example of IBX acid reaction
-
IBX oxidative cleavage
-
IBX DMSO oxidative cleavage
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian