Streptavidin
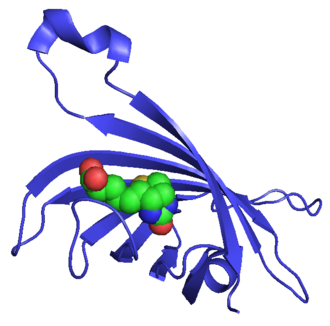



Streptavidin is a protein that is widely known for its strong affinity to the molecule biotin. It is a tetrameric protein produced by the bacterium Streptomyces avidinii. Streptavidin and biotin binding is one of the strongest non-covalent interactions in nature, making this interaction highly useful in a variety of biotechnology and molecular biology applications.
Structure[edit]
Streptavidin is composed of four identical subunits, each capable of binding a single biotin molecule. This tetrameric structure allows for a very tight binding to biotin with a dissociation constant (Kd) in the order of 10^-14 M, which is much lower than most other protein-ligand interactions. The protein has a relatively high molecular weight of approximately 60 kDa.
Applications[edit]
Due to its strong affinity for biotin, streptavidin is extensively used in the field of biotechnology and molecular biology. Some of the common applications include:
- Western Blot: Streptavidin is used to detect biotinylated proteins.
- ELISA (Enzyme-Linked Immunosorbent Assay): It is used to capture biotinylated antibodies or antigens.
- Immunohistochemistry: Streptavidin-biotin complex is used for staining specific proteins in tissue sections.
- Nucleic Acid Detection: Biotinylated DNA or RNA probes can be detected using streptavidin conjugated to fluorescent markers or enzymes.
- Purification Techniques: Streptavidin immobilized on a solid phase can be used to purify biotinylated compounds.
Advantages and Limitations[edit]
The streptavidin-biotin interaction is highly advantageous due to its unmatched strength and specificity. This allows for very stable complex formation that is resistant to changes in temperature, pH, and denaturing agents. However, the high affinity of streptavidin to biotin can also be a limitation, as it makes the dissociation of the complex difficult without denaturing the protein. This can be problematic in applications where the recovery of the biotinylated compound is desired.
Variants[edit]
Several engineered variants of streptavidin have been developed to overcome some of its limitations or to improve its utility in certain applications. These include:
- Streptavidin muteins: Mutations introduced to reduce the affinity to biotin, facilitating easier dissociation of the complex.
- Truncated streptavidin: Smaller versions of the protein that maintain biotin binding but have altered physical properties.
Conclusion[edit]
Streptavidin is a powerful tool in the arsenal of molecular biology and biotechnology, enabling a wide range of applications due to its strong and specific binding to biotin. Its use spans from basic research to diagnostic and therapeutic applications, highlighting its importance in the scientific community.

This article is a biochemistry stub. You can help WikiMD by expanding it!
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Get started with evidence based, physician-supervised
affordable GLP-1 weight loss injections
Now available in New York City and Philadelphia:
- Semaglutide starting from $59.99/week and up
- Tirzepatide starting from $69.99/week and up (dose dependent)
✔ Evidence-based medical weight loss ✔ Insurance-friendly visits available ✔ Same-week appointments, evenings & weekends
Learn more:
Start your transformation today with W8MD weight loss centers.
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian