Methyl red
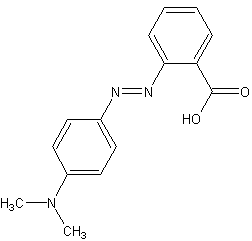
Methyl red (MR), also known as C.I. Acid Red 2, is an indicator dye that turns red in acidic solutions. It is a pH indicator used to identify the acidity of a solution. Methyl red is an azo dye, and it changes color at a pH of about 4.4 to 6.2, from red at pH 4.4 and below to yellow at pH 6.2 and above, with the transition range between these values appearing orange.
Chemistry
Methyl red is a chemical compound with the formula C15H15N3O2. It is part of the azo dye group, which are characterized by the presence of the functional group -N=N-, known as an azo group. In an acidic environment, methyl red offers a red color due to the presence of hydronium ions (H3O+) which stabilize the structure that absorbs at longer wavelengths. In neutral to basic conditions, the molecule exists in a form that absorbs at shorter wavelengths, thus appearing yellow.
Application
Methyl red is widely used in the laboratory as a pH indicator in titration experiments. It is particularly useful in acid-base titrations due to its clear and distinct color change. Besides its use in chemistry, methyl red is also employed in the microbiology field, specifically in the Methyl Red test, part of the IMViC tests, to identify bacteria that produce stable acid end products from glucose fermentation.
Methyl Red Test
The Methyl Red test is a procedure used in microbiology to detect the ability of microorganisms to perform mixed acids fermentation when supplied with glucose. The presence of acid end-products lowers the pH of the medium, and the addition of methyl red indicator to the culture turns the medium red, indicating a positive test. Organisms that utilize the butylene glycol pathway or other non-acidic pathways result in a negative test, with the medium turning yellow.
Safety
As with many azo dyes, there are safety considerations with methyl red. It should be handled with care in the laboratory, using appropriate personal protective equipment. Methyl red is considered to be a potential health hazard if ingested, inhaled, or comes into contact with skin and eyes.
Environmental Impact
The environmental impact of azo dyes, including methyl red, is a concern due to their synthetic nature and potential to release aromatic amines, some of which may be carcinogenic, upon degradation. Efforts are ongoing to develop more environmentally friendly alternatives and to improve the degradation processes of existing dyes to minimize their impact.
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
