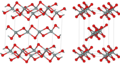
Silicon disulfide
Silicon disulfide is an inorganic compound with the chemical formula SiS2. It is a part of the group of compounds known as silicon sulfides, which also includes substances like silicon monosulfide (SiS) and silicon polysulfides (SiSx, where x > 2). Silicon disulfide is of interest both in materials science and in the study of the chemistry of silicon and sulfur.
Properties
Silicon disulfide exists as a solid under standard conditions. It is characterized by its high melting point and its resistance to oxidation compared to other silicon sulfides. The compound typically forms fibrous or amorphous powders, which can be reactive with water, releasing hydrogen sulfide (H2S) gas.
Chemical
The SiS2 molecule is composed of one silicon atom covalently bonded to two sulfur atoms. This configuration leads to a linear molecular geometry, similar to carbon disulfide (CS2). Silicon disulfide is a strong reducing agent and reacts with a variety of chemical reagents, including water, oxygen, and halogens.
Physical
Silicon disulfide is insoluble in water but can be dissolved in some organic solvents. It has a high melting point, indicative of the strong covalent bonds between the silicon and sulfur atoms. The compound's physical properties, such as density and refractive index, have been studied for their potential applications in materials science.
Synthesis
Silicon disulfide can be synthesized through several methods, including the direct reaction of elemental silicon with sulfur at high temperatures. Another method involves the reaction of silicon dioxide (SiO2) with carbon (C) and sulfur at high temperatures, producing SiS2 along with carbon monoxide (CO) as a byproduct.
Applications
While not extensively used in commercial applications, silicon disulfide has been studied for its potential use in high-temperature ceramics, as a precursor for the production of silicon-sulfur polymers, and in the development of batteries and electronic devices. Its unique properties, such as thermal stability and electrical conductivity, make it a compound of interest in advanced materials research.
Safety
Silicon disulfide should be handled with care, as it can decompose to release toxic hydrogen sulfide gas, especially when in contact with water. Proper safety equipment, such as gloves and eye protection, should be used when handling this compound. Additionally, adequate ventilation is necessary to prevent the accumulation of hydrogen sulfide gas in the air.
See Also
Silicon disulfide
-
SiS2 type Silica
-
SiS2 chain from Ibam xtal 2015 3D balls
-
SiS2
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Get started with evidence based, physician-supervised
affordable GLP-1 weight loss injections
Now available in New York City and Philadelphia:
- Semaglutide starting from $59.99/week and up
- Tirzepatide starting from $69.99/week and up (dose dependent)
✔ Evidence-based medical weight loss ✔ Insurance-friendly visits available ✔ Same-week appointments, evenings & weekends
Learn more:
Start your transformation today with W8MD weight loss centers.
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian