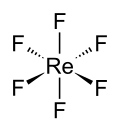
Rhenium hexafluoride
Rhenium hexafluoride is a chemical compound with the formula ReF6. It is one of the seventeen known binary hexafluorides. Rhenium hexafluoride is a dense, volatile, and highly reactive compound that is a colorless gas at room temperature, making it one of the most reactive and volatile compounds of rhenium. It is used in various chemical research applications and in the synthesis of other rhenium compounds.
Properties
Rhenium hexafluoride is a powerful oxidizing agent that can react violently with organic materials and is hydrolyzed by water to form oxygen and rhenium oxides. It has a high melting point and boils at temperatures significantly higher than room temperature, which makes it a gas under normal conditions. The compound has a complex electronic structure, with the rhenium atom exhibiting a +6 oxidation state, which is common for rhenium in its high oxidation state compounds.
Synthesis
The synthesis of rhenium hexafluoride typically involves the direct combination of rhenium with fluorine gas at high temperatures. This process requires careful control of reaction conditions to prevent the formation of unwanted byproducts and to ensure the purity of the ReF6 produced. The reaction is highly exothermic and must be conducted in apparatus made of materials that can withstand the corrosive effects of both reactants and the product.
Applications
Due to its potent oxidizing properties and volatility, rhenium hexafluoride is primarily used in scientific research. It serves as a fluorinating agent in the synthesis of other chemical compounds and can be used to introduce fluorine atoms into various molecular structures. Its applications are mainly confined to the laboratory due to its reactivity and the difficulty in handling the compound safely.
Safety
Handling rhenium hexafluoride requires strict safety precautions due to its high reactivity and toxicity. It can cause severe burns upon contact with skin and can be fatal if inhaled. Appropriate protective equipment, including gloves, goggles, and face shields, must be worn when working with this compound. Additionally, operations involving ReF6 should be conducted in a well-ventilated area or under a fume hood to avoid inhalation of fumes.
-
Rhenium(VI)-fluoride
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian