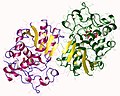
Bisphosphoglycerate mutase
Bisphosphoglycerate mutase (BPGM) is an enzyme that plays a crucial role in the glycolysis and gluconeogenesis metabolic pathways. It catalyzes the conversion of 1,3-bisphosphoglycerate (1,3-BPG) to 2,3-bisphosphoglycerate (2,3-BPG) and vice versa. This enzyme is predominantly found in the red blood cells where it facilitates the release of oxygen from hemoglobin to the tissues by stabilizing the deoxy form of hemoglobin. The presence and activity of BPGM are vital for the efficient functioning of the body's metabolic processes, especially under conditions of high altitude or hypoxia.
Function
BPGM has a critical function in the regulation of hemoglobin's oxygen-binding affinity. The enzyme's activity leads to the production of 2,3-BPG, which binds with greater affinity to deoxyhemoglobin than to oxyhemoglobin. This binding effect lowers the affinity of hemoglobin for oxygen, thereby facilitating the release of oxygen to the tissues. The regulation of 2,3-BPG levels by BPGM is essential for adapting to changes in oxygen availability, such as during physical exertion or at high altitudes.
Structure
The structure of bisphosphoglycerate mutase is complex, consisting of multiple domains that contribute to its catalytic activity. The enzyme operates as a dimer, with each subunit contributing to the overall function. The active site of BPGM is where the substrate binding and catalytic conversion occur, involving a series of coordinated chemical reactions.
Genetics
The gene responsible for encoding bisphosphoglycerate mutase is located on human chromosome 7. Mutations in this gene can lead to alterations in enzyme activity, which may impact oxygen delivery to tissues and can have clinical implications, including disorders related to red blood cell function and oxygen transport.
Clinical Significance
Alterations in BPGM activity can have significant clinical implications. For example, a decrease in enzyme activity can lead to reduced levels of 2,3-BPG, impairing the release of oxygen to tissues and leading to symptoms of hypoxia. Conversely, increased BPGM activity can enhance oxygen release, which may be beneficial in certain conditions but could also lead to issues such as tissue hyperoxia.
Research
Research into bisphosphoglycerate mutase continues to uncover its roles beyond oxygen regulation. Studies have explored its involvement in various diseases, potential as a therapeutic target, and its evolutionary significance in adapting to different environmental oxygen levels.

This article is a biochemistry stub. You can help WikiMD by expanding it!
Bisphosphoglycerate mutase gallery
-
2h4z
-
Whole Enzyme
-
Labeled Active Site
-
Coordination in Active site
-
Mechanism of 1-3BPG to 2-3BPG
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit.
- Tirzepatide starting from $45.00/week and up (dose dependent)
✔ Same-week appointments, evenings & weekends
Learn more:
Start your transformation today with W8MD weight loss centers.
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian