Woodward–Hoffmann rules: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 32: | Line 32: | ||
{{Chemistry-stub}} | {{Chemistry-stub}} | ||
<gallery> | |||
File:Stereospecificity_electrocyclic_ring_opening1.png|Woodward–Hoffmann rules | |||
File:Torquo.png|Woodward–Hoffmann rules | |||
File:Cyclobutenes.png|Woodward–Hoffmann rules | |||
File:WH_4n_thermal_MO.png|Woodward–Hoffmann rules | |||
File:WH_4n_photo_MO.png|Woodward–Hoffmann rules | |||
File:WH_4n+2_thermal_MO.png|Woodward–Hoffmann rules | |||
File:Symmetry_TransitionState.png|Woodward–Hoffmann rules | |||
File:WH_symmetry_of_MOs_4e_cyclization.png|Woodward–Hoffmann rules | |||
File:Symmetry_butadiene_MOs.png|Woodward–Hoffmann rules | |||
File:Symm_MOs_cyclobutene.png|Woodward–Hoffmann rules | |||
File:4e_conrot_correlationdiagram.png|Woodward–Hoffmann rules | |||
File:4e_disrot_correlationdiagram.png|Woodward–Hoffmann rules | |||
</gallery> | |||
Revision as of 11:03, 18 February 2025
Woodward–Hoffmann rules are a set of principles in organic chemistry and physical chemistry that predict the stereochemistry of the products of pericyclic reactions. These rules were formulated by Robert Burns Woodward, an American organic chemist, and Roald Hoffmann, an American theoretical chemist, in the early 1960s. The Woodward–Hoffmann rules are based on the conservation of orbital symmetry and provide a framework for understanding the behavior of electrons in a pericyclic reaction, which is a reaction that occurs in a single step, without intermediates, through a cyclic transition state.
Overview
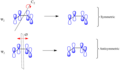
Pericyclic reactions are characterized by the reorganization of electrons and bonds, typically involving π electrons, through a cyclic transition state. The Woodward–Hoffmann rules help predict whether a pericyclic reaction will proceed under thermal or photochemical conditions by analyzing the symmetry properties of the molecular orbitals involved. According to these rules, a pericyclic reaction can proceed in a suprafacial or antarafacial manner, depending on the conservation of orbital symmetry.
Rules
The Woodward–Hoffmann rules can be summarized as follows:
- For thermal reactions:
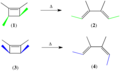
- Conrotatory motion is allowed for 4n π-electron systems in cycloaddition and electrocyclic reactions.
- Disrotatory motion is allowed for 4n+2 π-electron systems in electrocyclic reactions.
- For photochemical reactions:
- The allowed motions are reversed; disrotatory motion is allowed for 4n π-electron systems, and conrotatory motion is allowed for 4n+2 π-electron systems.
Applications
The Woodward–Hoffmann rules have wide applications in the synthesis of complex organic molecules. They are particularly useful in predicting the outcomes of Diels-Alder reactions, sigmatropic rearrangements, and other types of pericyclic reactions. Understanding these rules allows chemists to design synthetic pathways that lead to the desired stereochemistry of the product.
Significance
The development of the Woodward–Hoffmann rules was a significant milestone in the field of chemistry. It provided a theoretical foundation for understanding the behavior of electrons in pericyclic reactions, which was previously based on empirical observations. The rules have not only advanced the field of organic chemistry but also contributed to the development of new synthetic methods and materials. For their contributions, Woodward and Hoffmann were awarded the Nobel Prize in Chemistry in 1981.
See also
References
<references/>
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules
-
Woodward–Hoffmann rules