Zinc dithiophosphate: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 30: | Line 30: | ||
[[Category:Lubricants]] | [[Category:Lubricants]] | ||
{{Chem-stub}} | {{Chem-stub}} | ||
<gallery> | |||
File:Zn(dtp)2.png|Zinc dithiophosphate | |||
File:Zinc-diethyldithiophosphate-chain-from-xtal-3D-balls.png|Zinc dithiophosphate | |||
</gallery> | |||
Latest revision as of 01:42, 18 February 2025
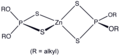
Zinc dithiophosphate (ZDDP) is an organosulfur compound with the formula (S2P(OC4H9)2)2Zn. It is a coordination complex consisting of zinc cations and dithiophosphate anions. ZDDP is a key additive in lubricating oils, providing anti-wear, antioxidant, and corrosion-resistant properties. It is widely used in the automotive industry, particularly in motor oil formulations, to protect engine parts from wear and tear.
Properties and Structure[edit]
ZDDP is a colorless to slightly yellowish fluid, with a characteristic odor. It is insoluble in water but soluble in oils and organic solvents. The compound forms by the reaction of phosphorus pentasulfide with alcohols, followed by neutralization with zinc oxide. The structure of ZDDP involves a zinc ion coordinated to the sulfur atoms of two dithiophosphate anions.
Applications[edit]
The primary application of ZDDP is as an additive in lubricants. It serves multiple functions:
- Anti-wear Agent: ZDDP forms a protective film on metal surfaces, reducing wear and extending the life of engine components.
- Antioxidant: It prevents the oxidation of the lubricant, thereby maintaining its quality and performance over time.
- Corrosion Inhibitor: ZDDP protects metal surfaces from corrosion caused by moisture and acidic compounds.
In addition to automotive lubricants, ZDDP is used in industrial oils, greases, and other lubricating formulations.
Environmental and Health Concerns[edit]
While ZDDP is effective in protecting engine parts, it has raised environmental and health concerns. The phosphorus component of ZDDP can lead to the formation of phosphorus oxides during combustion, contributing to air pollution. Moreover, excessive use of ZDDP can lead to the poisoning of automotive catalytic converters, reducing their efficiency in controlling exhaust emissions.
Regulatory Aspects[edit]
Due to its environmental impact, the use of ZDDP in lubricants is subject to regulatory limitations. Automotive manufacturers and oil companies are actively researching alternatives that provide similar protective qualities without the negative environmental effects.
See Also[edit]
References[edit]
<references/>
-
Zinc dithiophosphate
-
Zinc dithiophosphate