Hemocyanin: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import Tags: mobile edit mobile web edit |
||
| Line 28: | Line 28: | ||
{{Biochemistry-stub}} | {{Biochemistry-stub}} | ||
{{medicine-stub}} | {{medicine-stub}} | ||
== Hemocyanin == | |||
<gallery> | |||
File:Hemocyanin2.jpg|Hemocyanin2 | |||
File:PDB_1hcy_EBI.jpg|PDB 1hcy EBI | |||
File:PDB_1oxy_EBI.jpg|PDB 1oxy EBI | |||
File:Molluscan_hemocyanin_(4YD9).png|Molluscan hemocyanin (4YD9) | |||
File:deoxyhemocyanin_full.png|Deoxyhemocyanin full | |||
File:oxyhemocyanin_full.png|Oxyhemocyanin full | |||
File:Hemocyanin_Example.jpg|Hemocyanin Example | |||
</gallery> | |||
Latest revision as of 21:01, 23 February 2025
Hemocyanin is a protein that transports oxygen in the blood of many mollusks and some arthropods, such as the horseshoe crab (Limulus polyphemus). Unlike the hemoglobin found in vertebrates, hemocyanin is blue in color due to the presence of copper in its active site, which binds oxygen, giving the blood a distinctive blue color. Hemocyanin is a large molecule composed of multiple subunits, which can vary in number among different species.
Structure and Function[edit]
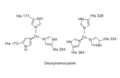
Hemocyanin operates on a principle similar to that of hemoglobin. Oxygen binding is reversible, and the oxygenation state of the molecule changes its color from dark blue when deoxygenated to a lighter blue when oxygenated. The oxygen-binding mechanism of hemocyanin involves the copper ions at its active site. Each copper ion can bind one oxygen molecule (O2), facilitating the transport of oxygen from the respiratory organs to the tissues that require it for metabolism.
The structure of hemocyanin is quite complex. It is made up of multiple subunits, which can be hexamers or dodecamers, forming a cylindrical shape. These subunits are themselves composed of functional units, each containing a pair of copper atoms. The quaternary structure of hemocyanin allows for efficient oxygen transport and has been a subject of extensive research to understand its evolutionary significance and functional mechanism.
Evolutionary Significance[edit]
Hemocyanin is of particular interest to scientists because it represents a different evolutionary solution to the problem of oxygen transport in the blood. While vertebrates developed hemoglobin, which uses iron to bind oxygen, some invertebrates developed hemocyanin, which uses copper. This difference is thought to reflect the varying availability of these metals in the ancestral environments of these organisms.
Clinical and Biotechnological Applications[edit]
Research into hemocyanin has also found clinical and biotechnological applications. For example, hemocyanin from the Chilean abalone (Concholepas concholepas) has been used as an immunostimulant in the treatment of bladder cancer. Its ability to trigger the immune response makes it a valuable tool in vaccine development and cancer therapy.
See Also[edit]
References[edit]
<references/>

This article is a biochemistry stub. You can help WikiMD by expanding it!
Hemocyanin[edit]
-
Hemocyanin2
-
PDB 1hcy EBI
-
PDB 1oxy EBI
-
Molluscan hemocyanin (4YD9)
-
Deoxyhemocyanin full
-
Oxyhemocyanin full
-
Hemocyanin Example