Phosphoinositide phospholipase C: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 26: | Line 26: | ||
{{stub}} | {{stub}} | ||
==Phosphoinositide_phospholipase_C== | |||
<gallery> | |||
File:Phospholipase_C-1.png|Phosphoinositide phospholipase C | |||
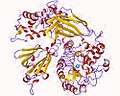
File:2zkm.jpg|Crystal structure of phospholipase C | |||
File:PIP2_cleavage_to_IP3_and_DAG.jpg|PIP2 cleavage to IP3 and DAG | |||
File:PDB_2fju_EBI.jpg|Structure of phospholipase C | |||
File:1djx_opm.png|Phosphoinositide phospholipase C | |||
</gallery> | |||
Latest revision as of 04:45, 18 February 2025
Phosphoinositide phospholipase C (PLC) is a family of enzymes that are known to cleave phospholipids just before the phosphate group. It plays a significant role in the intracellular signal transduction pathway.
Etymology[edit]
The term "Phosphoinositide phospholipase C" is derived from its function. "Phospho" refers to the presence of a phosphate group, "inositide" refers to the inositol ring structure, and "phospholipase C" refers to the specific type of enzyme that cleaves phospholipids.
Function[edit]
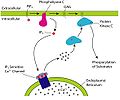
PLC is activated by G protein-coupled receptors and tyrosine kinase-linked receptors. Once activated, it cleaves Phosphatidylinositol 4,5-bisphosphate (PIP2) to form Inositol trisphosphate (IP3) and Diacylglycerol (DAG). IP3 is released into the cytosol where it binds to IP3 receptors, particularly in the endoplasmic reticulum (ER). This binding to the IP3 receptor stimulates the release of calcium ions (Ca2+) from the ER. DAG remains in the plasma membrane and acts as a secondary messenger, activating Protein kinase C (PKC).
Isoforms[edit]
There are thirteen known isoforms of PLC that are divided into six subfamilies depending on their structure and function. These include PLC-β, PLC-γ, PLC-δ, PLC-ε, PLC-ζ, and PLC-η.
Clinical significance[edit]
Mutations in PLC can lead to various diseases such as Usher syndrome, Leukemia, and Autoimmune lymphoproliferative syndrome.