Boyle's law: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import Tags: mobile edit mobile web edit |
||
| Line 36: | Line 36: | ||
{{physics-stub}} | {{physics-stub}} | ||
{{chemistry-stub}} | {{chemistry-stub}} | ||
== Boyle's law == | |||
<gallery> | |||
File:Boyles_Law_animated.gif|Boyles Law animated | |||
File:Boyles_Law.svg|Boyles Law | |||
File:Boyle's_Law_Demonstrations.webm|Boyle's Law Demonstrations | |||
</gallery> | |||
Latest revision as of 21:17, 23 February 2025
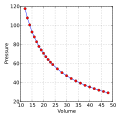
Boyle's Law is a fundamental principle in physics and chemistry, specifically within the sub-disciplines of physical chemistry and gas laws. Named after physicist and chemist Robert Boyle, this law describes the inversely proportional relationship between the absolute pressure and volume of a gas, if the temperature is kept constant within a closed system.
History[edit]
Robert Boyle (1627–1691), an Irish-born scientist who was a key figure in the 17th century scientific revolution, first stated the law in 1662. Boyle's experiments with gases led to the development of what we now know as Boyle's Law. His findings were published in the work New Experiments Physico-Mechanicall, Touching the Spring of the Air, and its Effects.
Statement of Boyle's Law[edit]
Boyle's Law can be stated as: At constant temperature for a fixed mass, the absolute pressure and the volume of a gas are inversely proportional. Mathematically, this can be represented as:
- P ∝ 1/V
- P = k(1/V)
where:
- P is the pressure of the gas.
- V is the volume of the gas.
- k is a constant value representative of the temperature and volume of the system.
Applications of Boyle's Law[edit]
Boyle's Law has numerous applications in real-life scenarios and various fields such as meteorology, human physiology, and engineering. For instance, it is used in calculating the air pressure change in scuba diving and understanding the behavior of gases in aerospace engineering.
See Also[edit]
References[edit]
<references />
Boyle's law[edit]
-
Boyles Law animated
-
Boyles Law
-
Boyle's Law Demonstrations