Sodium hydroxide: Difference between revisions
CSV import |
CSV import |
||
| Line 39: | Line 39: | ||
{{stub}} | {{stub}} | ||
<gallery> | |||
File:Sodium-hydroxide-crystal-3D-vdW.png|Sodium hydroxide crystal 3D model | |||
File:NaOH_-_drain-cleaner.jpg|Drain cleaner containing sodium hydroxide | |||
File:Paint_stripping_with_caustic_soda.jpg|Paint stripping with caustic soda | |||
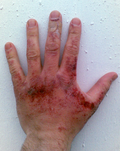
File:Sodium_hydroxide_burn.png|Sodium hydroxide burn | |||
File:NaOH_beads.jpg|Sodium hydroxide beads | |||
</gallery> | |||
Latest revision as of 05:02, 18 February 2025
Sodium Hydroxide (also known as caustic soda or lye) is an inorganic compound with the formula NaOH. It is a white solid ionic compound consisting of sodium cations Na+ and hydroxide anions OH−.
Chemical Properties[edit]
Sodium hydroxide is a highly caustic base and alkali that decomposes proteins at ordinary ambient temperatures and may cause severe chemical burns. It is highly soluble in water, and readily absorbs moisture and carbon dioxide from the air.
Production[edit]
Sodium hydroxide is industrially produced as a 50% solution by variations of the electrolytic Chloralkali process. Solid sodium hydroxide is obtained from this solution by the evaporation of water.
Uses[edit]
Sodium hydroxide is used in many industries, mostly as a strong chemical base in the manufacture of pulp and paper, textiles, drinking water, soaps and detergents and as a drain cleaner.
Safety[edit]
Sodium hydroxide is a dangerous chemical due to its ability to hydrolyze protein. If a dilute solution is spilled on the skin, it may not cause immediate pain, but it can lead to severe injury.
See Also[edit]
References[edit]
<references />
-
Sodium hydroxide crystal 3D model
-
Drain cleaner containing sodium hydroxide
-
Paint stripping with caustic soda
-
Sodium hydroxide burn
-
Sodium hydroxide beads
- Chemical compounds
- Industrial chemicals
- Alkalis
- Cleaning products
- Drain cleaners
- Desiccants
- Deliquescent substances
- Food additives
- Household chemicals
- Laboratory chemicals
- Photographic chemicals
- Reagents for organic chemistry
- Sodium compounds
- World Health Organization essential medicines
- Hydroxides
- All stub articles
- Medicine