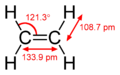Pi bond: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 30: | Line 30: | ||
[[Category:Molecular physics]] | [[Category:Molecular physics]] | ||
{{stub}} | {{stub}} | ||
<gallery> | |||
File:Ethylene_3D.png|Ethylene 3D structure | |||
File:Pi-Bond.svg|Pi bond illustration | |||
File:Ethane-staggered-CRC-MW-dimensions-2D.png|Ethane staggered conformation | |||
File:Ethylene-CRC-MW-dimensions-2D.png|Ethylene dimensions | |||
File:Acetylene-CRC-IR-dimensions-2D.svg|Acetylene dimensions | |||
</gallery> | |||
Latest revision as of 04:36, 18 February 2025
Pi bond
A pi bond (Greek: π) is a type of chemical bond that is characterized by the sharing of electrons between atoms in a molecule through p orbitals that are in contact through two areas of overlap. Pi bonds are usually weaker than sigma bonds.
Etymology[edit]
The term "pi bond" is derived from the Greek letter π, which is used in mathematics to represent a constant. In the context of chemistry, the term "pi bond" is used to describe a type of covalent bond that is formed by the overlap of p orbitals.
Formation[edit]
Pi bonds are formed when two atomic orbitals come together to form a molecular orbital that is symmetrical along the bond axis. The p orbitals that participate in pi bond formation are in a parallel orientation and must be unhybridized. The electrons in pi bonds are sometimes referred to as "pi electrons".
Properties[edit]
Pi bonds are generally weaker than sigma bonds due to the poor overlap of p orbitals. However, they are crucial in the formation of double and triple bonds in molecules. Pi bonds also play a significant role in the structures of organic molecules, such as alkenes and alkynes.