(+)-CPCA: Difference between revisions
No edit summary Tag: visualeditor-wikitext |
CSV import |
||
| (4 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
(+)-CPCA, also known as (+)-cis-3-phenylcyclopropylamine, is a chemical compound that acts as a monoamine oxidase inhibitor (MAOI). It is primarily of interest in the field of [[neuropharmacology]] due to its potential effects on [[neurotransmitter]] systems. | |||
== | ==Chemical Structure== | ||
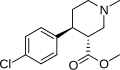
(+)-CPCA is a cyclopropylamine derivative, characterized by a three-membered cyclopropane ring attached to a phenyl group. The chemical formula is C9H11N, and it has a molecular weight of 133.19 g/mol. | |||
The | |||
==Mechanism of Action== | |||
As a monoamine oxidase inhibitor, (+)-CPCA functions by inhibiting the activity of the [[monoamine oxidase]] enzyme. This enzyme is responsible for the breakdown of monoamines, which are neurotransmitters such as [[serotonin]], [[dopamine]], and [[norepinephrine]]. By inhibiting this enzyme, (+)-CPCA increases the levels of these neurotransmitters in the brain, potentially affecting mood and behavior. | |||
==Potential Applications== | |||
Research into (+)-CPCA has primarily focused on its potential use in treating [[depression]] and other mood disorders. By increasing the availability of monoamines, it may help alleviate symptoms associated with these conditions. However, its use is limited by the potential for side effects and interactions with other medications. | |||
==Side Effects and Risks== | |||
The use of MAOIs, including (+)-CPCA, can lead to several side effects. Common side effects include dizziness, headache, and insomnia. More serious risks involve hypertensive crises, especially when combined with certain foods or other medications that increase monoamine levels. | |||
== | ==Research and Development== | ||
While (+)-CPCA has shown promise in preclinical studies, further research is needed to fully understand its efficacy and safety profile. Clinical trials are necessary to determine its potential as a therapeutic agent. | |||
{{nt}} | |||
[[Category:Monoamine oxidase inhibitors]] | |||
[[Category:Neuropharmacology]] | |||
[[Category:Experimental drugs]] | |||
{{ | <gallery> | ||
[[Category: | File:(+)-CPCA.svg|(+)-CPCA | ||
[[Category: | </gallery> | ||
[[Category: | |||
Latest revision as of 00:33, 20 February 2025
(+)-CPCA, also known as (+)-cis-3-phenylcyclopropylamine, is a chemical compound that acts as a monoamine oxidase inhibitor (MAOI). It is primarily of interest in the field of neuropharmacology due to its potential effects on neurotransmitter systems.
Chemical Structure[edit]
(+)-CPCA is a cyclopropylamine derivative, characterized by a three-membered cyclopropane ring attached to a phenyl group. The chemical formula is C9H11N, and it has a molecular weight of 133.19 g/mol.
Mechanism of Action[edit]
As a monoamine oxidase inhibitor, (+)-CPCA functions by inhibiting the activity of the monoamine oxidase enzyme. This enzyme is responsible for the breakdown of monoamines, which are neurotransmitters such as serotonin, dopamine, and norepinephrine. By inhibiting this enzyme, (+)-CPCA increases the levels of these neurotransmitters in the brain, potentially affecting mood and behavior.
Potential Applications[edit]
Research into (+)-CPCA has primarily focused on its potential use in treating depression and other mood disorders. By increasing the availability of monoamines, it may help alleviate symptoms associated with these conditions. However, its use is limited by the potential for side effects and interactions with other medications.
Side Effects and Risks[edit]
The use of MAOIs, including (+)-CPCA, can lead to several side effects. Common side effects include dizziness, headache, and insomnia. More serious risks involve hypertensive crises, especially when combined with certain foods or other medications that increase monoamine levels.
Research and Development[edit]
While (+)-CPCA has shown promise in preclinical studies, further research is needed to fully understand its efficacy and safety profile. Clinical trials are necessary to determine its potential as a therapeutic agent.
-
(+)-CPCA