1-Naphthylamine: Difference between revisions
CSV import |
CSV import |
||
| (One intermediate revision by the same user not shown) | |||
| Line 40: | Line 40: | ||
[[Category:Carcinogens]] | [[Category:Carcinogens]] | ||
[[Category:Chemical compounds]] | [[Category:Chemical compounds]] | ||
== 1-Naphthylamine == | |||
<gallery> | |||
File:1-Naphthylamine.png|1-Naphthylamine | |||
File:1-Naphthylamine-3D-balls.png|1-Naphthylamine 3D balls | |||
</gallery> | |||
== 1-Naphthylamine == | |||
<gallery> | |||
File:1-Naphthylamine.png|1-Naphthylamine structure | |||
File:1-Naphthylamine-3D-balls.png|1-Naphthylamine 3D ball model | |||
</gallery> | |||
Latest revision as of 00:42, 18 February 2025
1-Naphthylamine[edit]


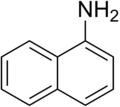
1-Naphthylamine is an organic compound with the formula C__H_NH_. It is one of the two isomeric naphthylamines, the other being 2-Naphthylamine. 1-Naphthylamine is a derivative of naphthalene, consisting of a naphthalene ring system with an amino group attached to the first carbon atom.
Structure and Properties[edit]
1-Naphthylamine is a crystalline solid that is typically colorless to pale yellow. It has a characteristic odor and is soluble in organic solvents. The compound exhibits basic properties due to the presence of the amino group, which can participate in hydrogen bonding and other interactions.
Synthesis[edit]
1-Naphthylamine can be synthesized through the reduction of 1-nitronaphthalene, which is itself derived from the nitration of naphthalene. The reduction process typically involves the use of iron and hydrochloric acid or other reducing agents.
Applications[edit]
1-Naphthylamine is used in the production of dyes, particularly azo dyes, where it serves as a precursor. It is also employed in the synthesis of various organic compounds and as an intermediate in the manufacture of pharmaceuticals.
Health and Safety[edit]
1-Naphthylamine is considered a hazardous substance. It is classified as a potential carcinogen, and exposure to it should be minimized. Proper safety measures, including the use of personal protective equipment and adequate ventilation, are essential when handling this compound.
Environmental Impact[edit]
Due to its potential health risks, the release of 1-naphthylamine into the environment is regulated. It can pose a threat to aquatic life and should be disposed of in accordance with environmental regulations.
Related Compounds[edit]
Related Pages[edit]
Gallery[edit]
-
Chemical structure of 1-Naphthylamine
-
3D model of 1-Naphthylamine
1-Naphthylamine[edit]
-
1-Naphthylamine
-
1-Naphthylamine 3D balls
1-Naphthylamine[edit]
-
1-Naphthylamine structure
-
1-Naphthylamine 3D ball model